Estimated reading time: 12 minutes
Key Takeaways
- Immunotherapy for pancreatic cancer is investigational for most patients and is not yet a standard part of routine care.
- Pancreatic tumors create a dense, immune-suppressive environment that blocks standard immune treatments from working effectively.
- Clinical trials are the primary way patients can access emerging immunotherapy approaches, including combination regimens, cancer vaccines, and checkpoint inhibitors.
- CAR-T therapy remains experimental for pancreatic cancer and is generally available only through specialized clinical trial centers.
- Molecular testing and pathology review are critical first steps for patients exploring trial eligibility, as many studies require specific biomarkers for enrollment.
- Research breakthroughs such as RNA vaccines targeting KRAS mutations and CD40-based combination strategies are generating early promise but require further study before becoming standard treatments.
- Supportive care and quality of life should remain central to every treatment plan, regardless of trial participation.
Table of contents
- What Immunotherapy for Pancreatic Cancer Is
- Why Immunotherapy Has Been Challenging in Pancreatic Cancer
- Clinical Trials Testing New Immunotherapy Approaches
- CAR-T Therapy and Other Cell-Based Treatments
- Current Research Breakthroughs in Pancreatic Cancer Immunotherapy
- Who May Be Eligible for Immunotherapy Trials
- How Patients and Families Can Evaluate Next Steps
- FAQ: Immunotherapy for Pancreatic Cancer
Introduction
Immunotherapy for pancreatic cancer is an investigational treatment approach that aims to help the immune system recognize and attack pancreatic cancer cells. For most patients, it is not yet a standard part of care. Ongoing clinical trials, CAR-T therapy studies, and other research breakthroughs are helping scientists test whether immune-based treatments can improve patient outcomes in the years ahead.
Pancreatic cancer has historically responded poorly to immunotherapy compared with cancers such as melanoma or certain lung cancers. This does not mean the field has stopped advancing. It means the science is evolving, and patients who want to explore these emerging therapies need clear, realistic information.
This article explains what is currently being studied, what patients and families should understand about investigational treatment options, and how to have an informed conversation with an oncology team.
1. What Immunotherapy for Pancreatic Cancer Is
Immunotherapy for pancreatic cancer is an investigational approach that helps the immune system recognize and attack pancreatic cancer cells. It works differently from other standard cancer treatments. Rather than destroying cancer cells directly, immune-based treatments activate or guide immune cells, especially T cells, so they can identify and attack the tumor.
To understand how immunotherapy fits into the treatment landscape, it helps to compare it with other approaches:
- Chemotherapy uses drugs that kill fast-growing cells throughout the body.
- Radiation delivers high-energy beams aimed at a targeted tumor area.
- Surgery physically removes the tumor when possible.
- Immunotherapy helps the immune system do the work of identifying and fighting cancer cells.
Pancreatic tumors are especially difficult to treat with this method. The disease often resists immune attack in ways that other cancers do not. Most current immunotherapy approaches for pancreatic cancer remain investigational, meaning they are not established as routine care and are mainly accessed through clinical trials.
Why Pancreatic Tumors Are Hard to Target
Pancreatic cancer develops a protective structure called the tumor stroma, which is a thick layer of supportive tissue that surrounds the cancer. This dense environment can physically block immune cells from reaching the tumor.
Beyond the physical barrier, the cancer can also suppress immune activity. This means the immune system becomes less effective even when it attempts to mount a response. Researchers describe these tumors as “cold,” meaning they have low levels of immune cell infiltration and tend to be poor candidates for standard immune checkpoint therapies.
This combination of physical shielding and immune suppression is one of the central challenges that research breakthroughs are working to solve. If you want a broader overview of how treatment, diagnosis, and support connect in pancreatic cancer care, see the guide to understand the disease in more detail.
2. Why Immunotherapy Has Been Challenging in Pancreatic Cancer
Immunotherapy for pancreatic cancer has not produced the same results seen in some other cancers, and the reasons are rooted in tumor biology. Pancreatic tumors often create a protective, immune-suppressive environment that blocks treatment response from reaching the cancer cells.
The tumor microenvironment refers to the cells, signals, blood vessels, and structural tissue that surround the tumor. In pancreatic cancer, this environment tends to be hostile to immune activity. It includes signals that actively tell immune cells to stand down and structural barriers that prevent them from penetrating the tumor.
This process is called immune evasion. Cancer cells develop ways to hide from or disable immune attack. The result is a “cold tumor,” which is a tumor with low immune cell infiltration that typically does not respond well to immune checkpoint inhibitors.
This is a key difference from “hot tumors” found in cancers like melanoma or certain lung cancers, where immune cells are already active inside the tumor and checkpoint inhibitors can release the brakes on that immune activity. Pancreatic cancer does not naturally offer that starting point for most patients.
Research breakthroughs in this area are focused on overcoming these biological barriers. Scientists are not expecting a single drug to solve the problem. Instead, they are building combination strategies designed to change the tumor environment and create an opening for immune treatment to work.
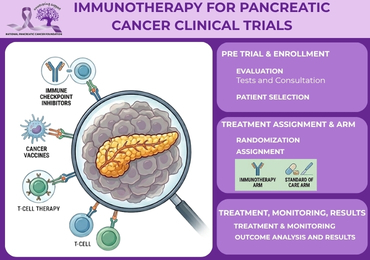
3. Clinical Trials Testing New Immunotherapy Approaches
Clinical trials are the primary way new immunotherapy for pancreatic cancer strategies are tested to determine whether they are safe and effective. Without clinical trials, none of the current advances in immune-based cancer treatment would have been possible.
Trials are organized into phases, each with a different purpose:
- Phase I focuses on safety, dosing, and identifying side effects in a small group of patients.
- Phase II looks at whether the treatment shows signs of working in a larger group.
- Phase III compares the new approach with standard care in a larger population to determine real-world benefit.
Trial designs can take different forms. Some use randomized comparisons between two groups. Others test combination therapies that pair immune treatments with chemotherapy or targeted agents. Some use “window of opportunity” designs that study immune effects before surgery or at the start of treatment in locally advanced disease.
Patients who participate in clinical trials may gain access to treatments that are not otherwise available. This can be meaningful in a disease where standard options are limited. Anyone interested in trial participation should speak with their oncologist, a clinical trial coordinator, or a specialist at a research hospital. A specialist center can also help patients compare treatment pathways, similar to what is covered in this resource on finding top care locations.
Examples of Trial Approaches Being Studied
Several research institutions are actively testing new immune-based combinations for pancreatic cancer. These examples show the range of strategies currently under investigation.
Researchers at UCLA have studied a virus-based immunotherapy combined with ezabenlimab, which is an immune checkpoint inhibitor, compared with standard care in patients with advanced pancreatic cancer.
UC San Diego has studied intratumoral mitazalimab, a CD40 antibody, delivered alongside electroporation for patients with locally advanced disease. CD40-targeted strategies are designed to stimulate immune response inside the tumor itself.
These are examples of the types of combinations researchers are testing. They are not proven standard treatments. They represent the investigational work happening inside the clinical trial system to determine what may help patients in the future. For readers who want a focused look at trial access and emerging therapies, the clinical trials guide may also be useful.
4. CAR-T Therapy and Other Cell-Based Treatments
CAR-T therapy is a personalized form of immunotherapy for pancreatic cancer in which a patient’s own T cells are collected, genetically modified, and returned to the body to fight cancer. It is one of the most closely watched areas of cancer research, but it remains experimental for pancreatic cancer and is generally available only through clinical trials.
Here is how the process works:
- T cells are collected from the patient’s blood.
- In a laboratory, those cells are genetically engineered to produce chimeric antigen receptors, also called CARs.
- These receptors are designed to recognize specific proteins on the surface of cancer cells.
- The modified T cells are multiplied in large numbers.
- They are then infused back into the patient, where they can seek out and attack the cancer.
CAR-T therapy is highly personalized, which makes it both promising and complex. Pancreatic cancer presents a particular challenge for this approach. Tumors can hide from immune cells, and they may express more than one protein target, making it easier for some cancer cells to escape attack.
Early studies have shown encouraging results. One reported disease control rate among responders reached 84.6% in a study examining engineered CAR-T cells targeting multiple antigens. However, this number does not mean most patients will see this result. These are early findings from clinical trials, not outcomes that apply broadly.
CAR-T therapy also carries potential risks. Cytokine release syndrome is one of the most serious, where a strong immune response causes system-wide inflammation. Treatment requires specialized centers with experience managing these reactions, which limits availability.
Other Cell-Based Approaches
Beyond CAR-T, researchers are also exploring multi-antigen adoptive T-cell therapies. These strategies target more than one cancer marker at the same time.
The rationale is practical. When a therapy targets only one antigen, pancreatic cancer cells that do not express that antigen can survive and continue growing. Targeting multiple antigens at once reduces the chance that the tumor escapes by simply changing one surface protein.
These approaches are still in early-stage research. They are part of a broader effort to improve immune recognition in tumors that have proven especially hard to treat.
5. Current Research Breakthroughs in Pancreatic Cancer Immunotherapy
Research breakthroughs in immunotherapy for pancreatic cancer typically refer to early-stage findings, not proven cures. It is important for patients and families to understand that “breakthrough” in research terms means a promising development that requires further study, not a treatment that is ready for routine use.
With that context in mind, several areas of research are generating significant interest.
Combination therapy is the dominant strategy. Researchers are pairing immunotherapy with chemotherapy, targeted therapy, radiation, or other immune agents. The goal is to use one treatment to open the door and another to walk through it.
RNA vaccines targeting KRAS mutations represent one of the most discussed advances in recent years. KRAS mutations are found in the vast majority of pancreatic cancers, making them an attractive target. Early findings have included some patients surviving beyond two years, which has drawn attention from the research community.
CD40 agonists such as mitazalimab are being studied in combination with chemotherapy. CD40 is a protein that can activate immune cells when targeted, potentially helping the immune system mount a stronger response against the tumor.
Triple immunotherapy approaches blocking checkpoint proteins 41BB and LAG3, combined with other immune modulators, have shown tumor regression in research settings. These multi-target strategies reflect the understanding that one checkpoint alone is rarely enough.
Tumor microenvironment targeting is another active research direction. Agents that block CXCR4, a protein that helps create a protective barrier around the tumor, and TTX030, which inhibits CD39, are being studied in combination with immunotherapy to change the environment inside the tumor and make it more vulnerable to immune attack.
Biomarker-driven approaches are also shaping trial design. Treatments are being tailored to specific tumor features such as claudin 18.2 expression, MSI-high status, KRAS mutation type, and DNA repair defects. This precision oncology approach is helping researchers match patients with the trials most likely to benefit them.
Johns Hopkins investigators have released a free atlas of immunotherapy responses in pancreatic cancers and reported new clinical trial findings, contributing to the field’s growing body of knowledge about how and why some tumors respond and others do not.
CAR-T therapy trials are also contributing to this picture, with multi-antigen strategies offering insights into how cell-based treatments might be refined for greater effectiveness.
6. Who May Be Eligible for Immunotherapy Trials
Eligibility for a pancreatic cancer immunotherapy clinical trial depends on several factors. There is no single answer that applies to all patients because each trial is designed with specific criteria for who can participate.
Common eligibility factors include:
- Cancer stage (early, locally advanced, or metastatic)
- Prior treatment history and what therapies have already been used
- Overall health and performance status, which reflects how well a patient can carry out daily activities
- Specific tumor characteristics identified through molecular testing or pathology review
- Biomarker results, which may include MSI-high status, KRAS mutation type, BRCA1 or BRCA2 mutations, PALB2 alterations, claudin 18.2 expression, or homologous repair deficiencies
Some trials are designed specifically for patients with advanced or metastatic disease. Others focus on locally advanced tumors or even earlier-stage patients in “window of opportunity” designs before surgery.
Patients interested in trial enrollment should ask their oncologist or clinical trial coordinator about trial matching services. These services review a patient’s profile against open studies to identify potential fits. For families also evaluating the broader treatment path, a general treatment overview can help put trial options in context.
Why Molecular Testing Matters
Molecular testing is one of the most practical first steps a patient can take when exploring clinical trial options. It can reveal features of the tumor that are not visible through standard imaging or basic pathology.
Genetic changes such as specific KRAS mutations, DNA repair defects, or other molecular markers may open doors to targeted or immune-based clinical studies that would not otherwise be available. Some trials require a specific biomarker as an entry criterion, meaning patients without that molecular result would not qualify.
A pathology review confirms the diagnosis and may identify additional tumor markers that are relevant to trial matching. Taken together, molecular testing and pathology review form the foundation of an informed conversation about investigational treatment options.
7. How Patients and Families Can Evaluate Next Steps
Patients who want to explore immunotherapy for pancreatic cancer should start by asking their oncology team whether molecular testing or a clinical trial review could identify investigational options that match their situation. This conversation is best started early, before treatment paths become more limited.
Practical questions to bring to an oncologist or clinical trial coordinator include:
- What clinical trials are currently open and match my tumor profile?
- What phase is the trial, and what does that mean for what is known so far?
- What are the possible benefits and risks of participating?
- What side effects should we expect, and how are they managed?
- Does this study require travel, extra appointments, or specialized facilities?
- How might participation affect quality of life during and after treatment?
- Would a second opinion at a research hospital help identify additional options?
Families play an important role in this process. Weighing the potential benefit of an investigational treatment against the added burden of trial participation is a personal decision. Extra visits, more testing, and the uncertainty of whether a new treatment will help are all real factors. So is the possibility of accessing a therapy that would not be available outside of a study.
Supportive care should be part of every treatment plan, regardless of whether a patient is in a trial. Symptom control, nutrition, pain management, and emotional support all contribute to quality of life and can affect how well a patient tolerates treatment. Guidance on living well through treatment can also be found in this support resource.
The National Pancreatic Cancer Foundation offers resources and guidance for patients and families navigating these decisions.
Research breakthroughs in this field are most meaningful when patients have access to informed decision-making support. Connecting with the right oncology team, asking the right questions, and understanding what investigational treatment actually means are the most important steps a family can take.
FAQ: Immunotherapy for Pancreatic Cancer
What is the latest immunotherapy for pancreatic cancer?
There is no single approved immunotherapy that works for most pancreatic cancer patients. The most current advances are found in clinical trials testing combination regimens, cancer vaccines, checkpoint inhibitors, and cell-based treatments like CAR-T therapy. Some studies focus on biomarker-guided approaches or strategies designed to change the tumor microenvironment and improve immune response. These are investigational approaches, not routine standard treatments.
Is immunotherapy effective for pancreatic cancer?
For most patients, immunotherapy has not been highly effective as a standalone treatment. The disease creates biological barriers that limit immune response. Some patients with specific biomarkers, such as MSI-high status, or those participating in clinical trials may see benefit, but overall response rates remain limited. Researchers are actively studying combination regimens and tumor-microenvironment strategies to improve these outcomes.
Are there any clinical trials for pancreatic cancer immunotherapy?
Yes, multiple clinical trials are currently testing immunotherapy combinations, vaccines, checkpoint inhibitors, and cell-based therapies for pancreatic cancer. Trial availability depends on a patient’s location, cancer stage, and tumor characteristics. An oncologist or clinical trial coordinator at a research hospital can help identify open studies that may be a fit. Patients can also explore options through institutional trial listings at major cancer centers.
Is CAR-T therapy available for pancreatic cancer?
CAR-T therapy is not a standard treatment for pancreatic cancer. It remains experimental and is generally available only through clinical trials at specialized centers. Patients interested in CAR-T should ask their oncologist whether any recruiting trials are currently open and whether they meet the eligibility criteria to participate.
What research breakthroughs are there for pancreatic cancer?
Scientists are currently testing combination immunotherapy regimens, RNA vaccines targeting KRAS mutations, CD40-based treatments, biomarker-guided therapy, and strategies that target the tumor microenvironment to make tumors more responsive to immune attack. These are promising early findings, not proven cures. Research breakthroughs in this context mean encouraging results from studies that are still working toward clinical confirmation. Johns Hopkins investigators have also published a free atlas of immunotherapy responses in pancreatic cancers, and additional early findings are emerging from trials at institutions such as UC San Diego.
Who qualifies for immunotherapy clinical trials?
Eligibility depends on cancer stage, prior treatment history, overall health status, and specific tumor biomarkers. Molecular testing and pathology review often help determine whether a patient fits the criteria for a particular study. A clinical trial coordinator or oncologist can walk through current options and assist with the screening process.
Moving Forward: What Patients and Families Should Know
Immunotherapy for pancreatic cancer remains investigational for most patients, but the field is advancing steadily through clinical trials and research breakthroughs that did not exist a decade ago. CAR-T therapy and other cell-based treatments show early promise, even as they remain experimental and trial-only for now.
The most practical step any patient can take is to ask their oncology team about molecular testing, clinical trial matching, and whether a referral to a research center is appropriate for their situation. Families should weigh the potential opportunity of investigational treatment against the real demands of trial participation, and they should make sure supportive care and quality of life remain central to every plan.
The National Pancreatic Cancer Foundation provides resources to help patients and families stay informed and connected to the support they need throughout this process. Staying current with emerging research and working closely with a knowledgeable oncology team remain the most important steps any family can take.